Services
The Advanced Light Microscopy Facility provides training and access to advanced light microscopy systems at an hourly rate. In addition, we are available to consult with and support users at every stage of a project including: experimental design, sample preparation, image acquisition, analysis, and data preparation.
- Consultation: We are available to meet with current and potential users to advise on sample preparation and the selection of the instrument that best serves the research requirements.
- Training: All users are provided with training that covers facility policies and details of instrument operation.
- Instrument Access: Researchers are able to independently use the instruments after training, or they can work with the Director in assisted sessions. See Instrumentation for current rates.
- Support: Our expertise is always available to help maximize the impact of the instruments on users’ studies through further consultations or by scheduling assisted sessions with the Director on the microscope.
- Education: The Facility gives workshops and lectures on microscopy applications and image analysis to advance the microscopy knowledge of the research community.
- Billing: Usage is tabulated and billing is executed by the Center for Open Research Resources and Equipment (COR2E).
Abberior Instruments 3D STED
This is an inverted confocal microscope that uses stimulated emission depletion (STED) of fluorescent dyes to obtain super resolved images. There are four pulsed excitation lasers and two pulsed STED depletion lasers for imaging. The instrument is capable of 2D or 3D super resolution imaging using spatial light modulators to shape the depletion beams in the x-y and z axes. The instrument is housed within the Bioscience Electron Microscopy Laboratory located in the Biology Physics Building room G11.
This microscope was acquired with a NIH shared instrumentation grant. Manuscripts including data from the Abberior STED system must include an acknowledgement of NIH grant S10OD023618 awarded to Christopher O’Connell.
Specifications
Microscope:
- Inverted Stand Olympus IX83
- Motorized x-y stage
- Physik Instrumente piezo z stage insert
Lasers:
Excitation
- 405 nm CW (non-STED imaging)
- 440 nm pulsed
- 485 nm pulsed
- 561 nm pulsed
- 640 nm pulsed
STED Depletion
- 595 nm pulsed
- 775 nm pulsed
Scanner:
- Continuously adjustable QUADScan galvo scanner (up to 2600 Hz line scanning)
Objective Lenses:
- 10X/0.3 UPLFLN10X2
- 20x/0.75 UPLSAPO20
- 40x/0.1.3 UPLFNL40 oil immersion
- 60x/1.2 UPLSAPO60 water immersion
- 100x/1.40 UPLANSAPO oil immersion
Detectors:
- Four filter-based APD detectors
Software:
- Abberior Impsector for acquisition and visualization
| Internal | External |
|
| Unassisted Use | $22.80/hour | Contact |
| Assisted Use | $89.80/hour | Contact |
| Instrument Training | $89.80/hour | Contact |
Leica SP8 Spectral Confocal
This is an inverted confocal microscope with five filter-free spectral and individually regulatable channels including four standard PMT detectors and one high-sensitivity HyD detector with photon counting capacity. The scanner also includes both standard galvo scan mirrors and an optional 8 KHz resonant scanner for high speed imaging. Use of an acusto-optical beamsplitter (AOBS) instead of traditional dichroic mirrors makes this microscope also suitable for reflected light microscopy applications. The instrument is housed in the Bioscience Electron Microscopy Laboratory located in the Biology Physics Building room G10.
This microscope was acquired with a NIH shared instrumentation grant. Manuscripts including data from the Leica SP8 confocal should include an acknowledgement of NIH grant S10OD016435 awarded to Akiko Nishiyama.
Specifications
Microscope:
- Inverted Stand DMI 6000
- Motorized stage
- Super Z Galvo stage
Lasers:
- 405 nm Diode laser
- Argon/2 (458, 488, 514 nm)
- 561 nm DPSS laser
- 633 nm HeNe laser
Scanners:
- Continuously adjustable non-resonant scanner (1-1800 Hz)
- Resonant scanner (8000 Hz) – 28 fps at 512 x 512
Objective Lenses:
- 10x/0.30 HC PL FLUOTAR
- 20x/0.75 HC PL APO IMM CORR CS2
- 40x/1.30 HC PL APO OIL CS2
- 63x/1.40 HC PL APO OIL CS2
- 100x/1.40 HCX PL APO OIL CS
Detectors:
- Four PMTs
- One HyD
- All filter-free spectral and individually regulatable detectors
- Transmitted light brightfield detector (with DIC option)
Software:
- LAS X for image acquisition, processing, and quantification
| Internal | External | |
| Unassisted Use | $21.60/hour | Contact |
| Assisted Use | $88.60/hour | Contact |
| Instrument Training | $88.60/hour | Contact |

Leica Thunder Imager
The Leica Thunder Imager is an upright microscope for fluorescence, transmitted light, and true color imaging . With a stage capable of holding up to 8 slides, this instrument is optimized for high throughput imaging of cells and tissue sections. Fluorescent images can be processed with Leica’s computational clearing and deconvolution to remove out of focus blur, enhance contrast, and sharpen details. The software Navigator function can be used for rapidly generating sample overviews for subsequent selection of areas of interest. The instrument is located in the Biology Physics Building room G05D.
Specifications
Microscope:
- Upright Stand Leica DM6 B
- Motorized x-y stage with 8 slide holder
Fluorescent Illuminator:
- LED3 solid state white light source
Fluorescent Filter Cubes
- DAPI: Ex 405/60, Em 470/40
- GFP: Ex 470/40, Em 525/50
- Y3: Ex 545/25, Em 605/70
- Y5: Ex 620/60, Em 700/75
Cameras:
- Leica K5 sCMOS monochrome camera, 2048 x 2048 pixels (for fluorescence imaging)
- Leica DMC5400 CMOS color camera (for histology dyes)
Objective Lenses:
- 2.5x/0.07 Plan
- 5x/0.15 Plan Fluotar
- 10x/0.45 Plan Apo
- 20x/0.80 Plan Apo, DIC
- 40x/1.30 Plan Fluotar Oil, DIC
- 63x/1.40 plan Apo Oil, DIC
- 63x/1.30 Plan Apo Glycerol
Software:
- LAS X for image acquisition, processing, and quantification. Leica Thunder software module includes Instant Computation Clearing (ICC) Small Volume Computation Clearing (SVCC) and Large Volume Computational Clearing (LVCC)
| Internal | External | ||
| Unassisted Use | $11.40/hour | Contact | |
| Assisted Use | $78.40/hour | Contact | |
| Instrument Training | $78.40/hour | Contact |

Nikon AXR Confocal + TIRF
The Nikon AXR is a point scanning confocal built around the inverted Ti2E microscope. The large 25 mm field of view and resonant scanner are optimized for rapid imaging of samples. The DUX-VB confocal detector features 4 sensitive GaAsP detectors that can be flexibly configured for a range of dyes via spectral detection. 6 laser lines provides a range of excitation options for a variety of fluorescent dyes. In addition to confocal microscopy, the system has an iLAS2 illumination module for TIRF imaging. This device can perform simultaneous TIRF imaging and photostimulation. TIRF, widefield, and transmitted light images are collected on a Photometrics Prime 95B sCMOS camera. The instrument is located in the Biology Physics Building room G05D.
Specifications
Microscope:
- Inverted microscope Nikon Ti2E
- Motorized x/y stage
- PerfectFocus optical z drift correction
- MCL Nano-Z200 piezo z stage
Laser Unit for Confocal:
- LUA-S6 405/445/488/514/561/640
Laser Unit for TIRF:
- LUNF XL 405/488/561/640
TIRF/Widefield Camera:
- Photometrics Prime95B
Objective Lenses:
- 10X PLAN APOCHROMAT LAMBDA D, DIC
- 20X PLAN APOCHROMAT LAMBDA D, DIC
- 40X/1.30 PLAN FLUOR, Oil, DIC
- 60X/1.40 PLAN APOCHROMAT LAMBDA D, Oil, DIC
- 100X/1.45 PLAN APOCHROMAT, Oil, DIC
- 100X/1.49 APO TIRF, Oil, DIC
Software:
- NIS-Elements C Confocal Package, JOBS for Scanning Plates and Creating Custom Acquisition and Analysis Routines, NIS-Elements 2D/3D, Deconvolution Suite, NIS.ai Artificial Intelligence Module, General Analysis 2 and 3
| Internal | External | |
| Unassisted Use | $21.60/hour | Contact |
| Assisted Use | $88.60/hour | Contact |
| Instrument Training | $88.60/hour | Contact |

Zeiss Lightsheet 7
The Zeiss Lightsheet 7 microscope is designed for fast, gentle 3D/4D imaging of samples labeled with fluorescent probes. Both live and fixed samples can be imaged. Samples are lowered from above into an imaging chamber containing media of the appropriate refractive index. It is located in the Biology Physics Building room G05.
Specifications
Lasers:
- 405, 488, 561, 638 nm
Imaging Optics:
- 5X 0.16NA (for live samples in aqueous media)
- 5X 0.16 NA (for fixed samples, correction collar adjustable for different refractive index media up to RI=1.53)
- 10X NA 0.5 (for live samples in aqueous media)
- 20X NA 1.0 (for live samples in aqueous media)
- 20X NA 1.0 (for fixed samples, correction collar adjustable for different refractive index media up to RI=1.45)
Illumination Optics (2 each for forming the lightsheet):
- 5X NA 0.1
- 10X NA 0.2
Imaging Chambers:
- Live chamber for samples in aqueous media
- Fixed sample chamber (small)
- Fixed sample chamber (large, 28 x 31 x 49 mm)
Detectors:
- 2 PCO edge 4.2 sCMOS cameras for dual channel imaging
- 2048 x 2048 pixel resolution
- 100 fps maximum frame rate
- up to 82 % quantum efficiency
Software:
- Zen Black for acquisition
- Zen Blue for analysis
- Arivis for large volume 3D rendering and analysis (on separate analysis PC)
| Internal | External |
|
| Unassisted Use | $17.40/hour | Contact |
| Assisted Use | $84.40/hour | Contact |
| Instrument training | $84.40/hour | Contact |

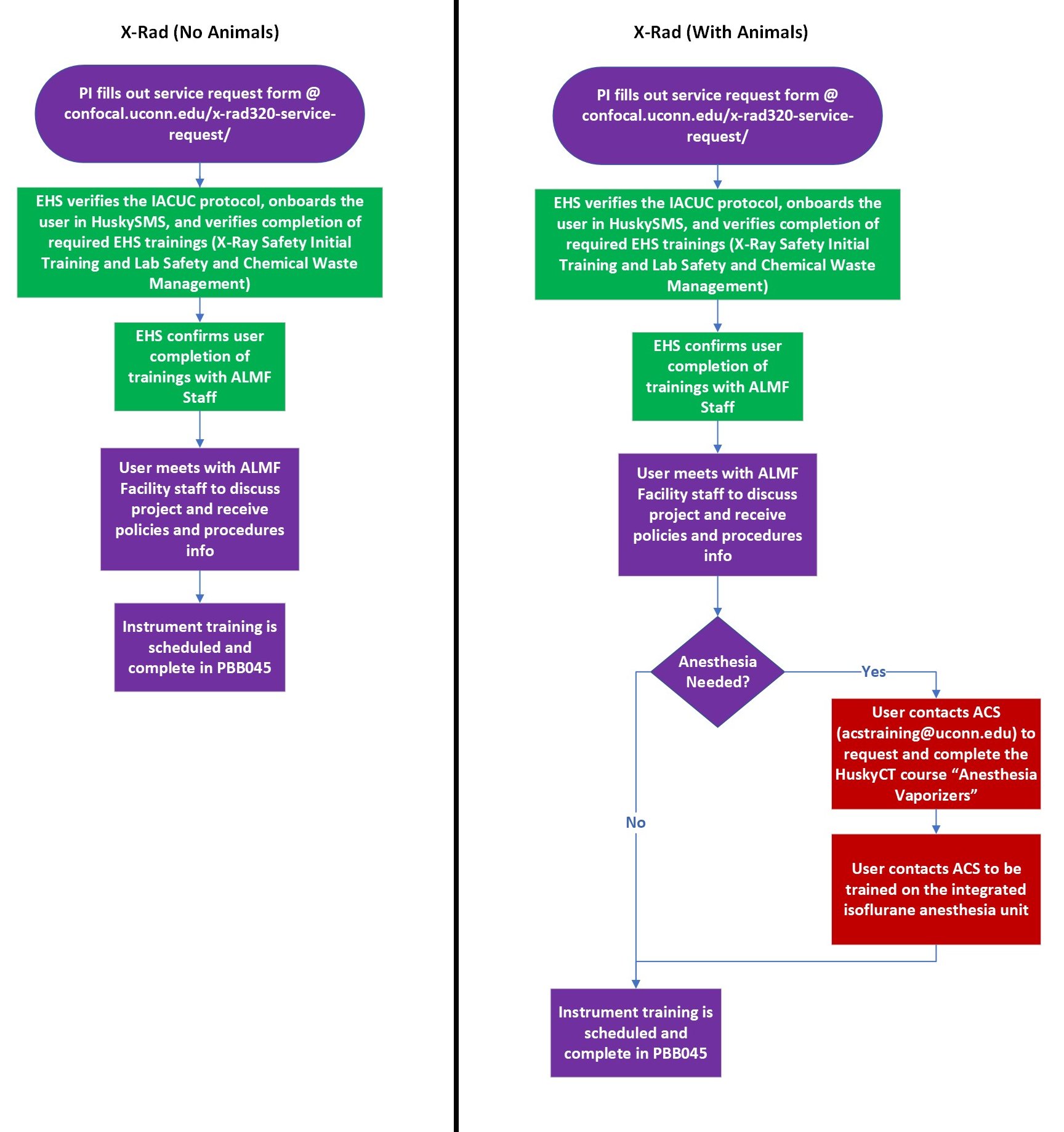
X-Rad320 Irradiator
The Precision X-Rad320 Irradiator delivers targeted x-ray radiation to a wide range of specimens. The optional Optimax imaging mode can be used for x-ray, bioluminescence, or optical imaging. Researchers wanting to use this instrument should refer to the below flowcharts. These outline the procedures for gaining access to the instrument. Access, training, and usage are coordinated by the Advanced Light Microscopy Facility and includes involvement of UConn Environmental Health and Safety (EHS) and Animal Care Services (ACS) when relevant.
Specifications
- PIs must submit the dedicated X-Rad320 Service Request Form for any researchers in their labs who will use this instrument.
- Only researchers who are going to actively perform research with the instrument will be authorized to access the room with the X-Rad320 Irradiator.
| Internal | External |
|
| Unassisted Use | $46.00/hour | Contact |
| Assisted Use | $113.00/hour | Contact |
| Instrument training | $113.00/hour | Contact |

Image Analysis Workstation
The Image Analysis Workstation is a powerful computer dedicated for image analysis and visualization. Several software packages are installed to support a variety of data formats.
Specifications
Location:
Bioscience Electron Microscopy Laboratory. Biology Physics Building room G10.
Specifications:
Hardware:
- HP Z640 Workstation
- 30″ Z30i LED-LCD Monitor
- Microsoft Windows 7 Professional Edition 64bit OS
- NVIDIA Quadro K2200 4GB Graphics Card
- Intel® Xeon E5-1660v3 3.00GHz 20MB 2133 8C CPU
- 64GB RAM
- 2 TB striped RAID data drive
Software:
- Leica LAS X including the following modules:
- 2D Analysis, 3D Analysis, and 3D Visualization
- NIS-Elements AR including the following module:
- Deconvolution
- Image J
Internal Rates: No charge for Confocal Microscopy Facility Users. A calendar is maintained for reserving time on the workstation.